Endpoint Health´s Challenge
Early-stage biopharma company in need of a product development team for their pre-clinical-stage drug-device combination approach for an immune mediated disorder.
With Adnovate we instantly created a turn-key clinical development function in the company. The Adnovate team functioned as an extension of the Endpoint internal team supporting clinical and non-clinical activities. Every member of the Adnovate team was of top quality and served as a model for what we would later look for when recruiting our own internal team.
Diego Rey | CSO Endpoint Health.
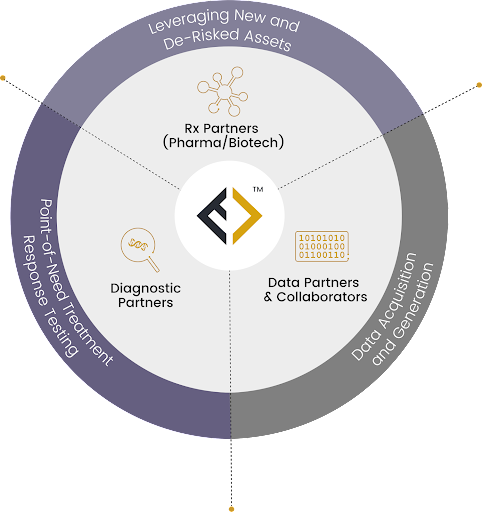
Adnovate Clinical’s Solution
Prior to the creation of their internal development function, Endpoint Health (EPH) partnered with Adnovate Clinical to provide functional leadership through provision of an interim CMO and staff to support all clinical development functions on an ‘as needed’ and flexible basis. Throughout this period Adnovate Clinical designed and guided the development of the clinical and regulatory plan for a phase 2 study using a companion diagnostic together with the therapeutic in collaboration with the internal company team, ensuring alignment with all major regulatory concepts, identifying key emerging risks and offering mitigation strategies. Adnovate Clinical produced a high-quality protocol designed with the agreement of a scientific and clinical advisory board, incorporated innovative trial methodology including PK/PD modelling and statistical methods and prepared the strategy, briefing documents, and executed the supporting work for a pIND interaction with the FDA to help meet corporate milestones. Adnovate Clinical continue to support EPH with the development of other assets.
;)